1-Propanol
Skeletal formula of 1-propanol
Ball and stick model of 1-propanol
Names
Pronunciation
Preferred IUPAC name
Other names
n -Propyl alcoholn -Propanoln -PrOHEthyl carbinol 1-Hydroxypropane Propionic alcohol Propionyl alcohol Propionylol Propyl alcohol Propylic alcohol Propylol
Identifiers
3D model (JSmol)
Beilstein Reference
1098242
ChEBI
ChEMBL
ChemSpider
DrugBank
ECHA InfoCard
100.000.679
EC Number
Gmelin Reference
25616
KEGG
MeSH
1-Propanol
RTECS number
UNII
UN number
1274
InChI=1S/C3H7OH/c1-2-3-4/h4H,2-3H2,1H3
Y Key: BDERNNFJNOPAEC-UHFFFAOYSA-N
Y
Properties
C 3 H 8 O
Molar mass
−1
Appearance
Colorless liquid
Odor
mild, alcohol-like[ 2]
Density
0.803 g/mL
Melting point
−126 °C; −195 °F; 147 K
Boiling point
97 to 98 °C; 206 to 208 °F; 370 to 371 K
Solubility in water
miscible
log P
0.329
Vapor pressure
1.99 kPa (at 20 °C)
Acidity (pK a )
16
Basicity (pK b )
−2
−45.176·10−6 cm3 /mol
1.387
Viscosity
1.959 mPa·s (at 25 °C)[ 3]
1.68 D
Thermochemistry
143.96 J/(K·mol)
Std molar(S ⦵ 298 )
192.8 J/(K·mol)
Std enthalpy of(Δf H ⦵ 298 )
−302.79…−302.29 kJ/mol
Std enthalpy of(Δc H ⦵ 298 )
−2.02156…−2.02106 MJ/mol
Pharmacology
D08AX03 (WHO
Hazards
Occupational safety and health
Main hazards
Flammable liquid
GHS labelling:
Pictograms
Signal word
Danger
Hazard statements
H225 , H302 , H318 , H336
Precautionary statements
P210 , P261 , P280 , P305+P351+P338
NFPA 704
Flash point
22 °C (72 °F; 295 K)
Autoignition
371 °C (700 °F; 644 K)
Explosive limits
2.2–13.7%[ 2]
Lethal dose or concentration (LD, LC):
LD50 (median dose)
2800 mg/kg (rabbit, oral)[ 4]
NIOSH (US health exposure limits):
PEL (Permissible)
TWA 200 ppm (500 mg/m3 )[ 2]
REL (Recommended)
TWA 200 ppm (500 mg/m3 ) ST 250 ppm (625 mg/m3 ) [skin][ 2]
IDLH (Immediate danger)
800 ppm[ 2]
Related compounds
Related compounds
Propane Isopropyl alcohol Ethanol
Supplementary data page
1-Propanol (data page)
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
1-Propanol (also propan-1-ol , propanol , n-propyl alcohol ) is a primary alcohol with the formula CH3 CH2 CH2 OH and sometimes represented as PrOH or n -PrOHisomer of 2-propanol . 1-Propanol is used as a solvent in the pharmaceutical industry, mainly for resins and cellulose esters, and, sometimes, as a disinfecting agent.
History
The compound was discovered by Gustave Chancel in 1853 by fractional distillation of fusel oil.[ 5] propionic alcoholhydrate of trityl .[ 6] [ 7]
After several unsuccessful attempts, it was synthesized independently and by two different routes by Eduard Linnemann and Carl Schorlemmer in 1868.[ 8] [ 9] [ 10]
Occurrence
Fusel alcohols like 1-Propanol are grain fermentation byproducts, and therefore trace amounts of 1-Propanol are present in many alcoholic beverages.
Chemical properties
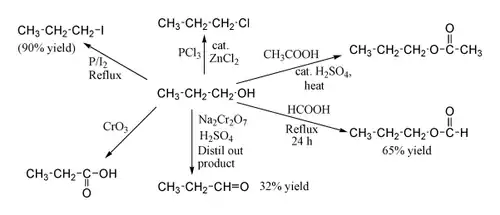
Some example reactions of 1-propanol 1-Propanol shows the normal reactions of a primary alcohol. Thus it can be converted to alkyl halides ; for example red phosphorus and iodine produce n-propyl iodide in 80% yield, while PCl3 with catalytic ZnCl2 acetic acid in the presence of an H2 SO4
Oxidation of 1-propanol with Na2 Cr2 O7 and H2 SO4 gives a 36% yield of propionaldehyde, and therefore for this type of reaction higher yielding methods using PCC or the Swern oxidation are recommended. Oxidation with chromic acid yields propionic acid .
Preparation
1-Propanol is manufactured by catalytic hydrogenation of propionaldehyde. Propionaldehyde is produced via the oxo process by hydroformylation of ethylene using carbon monoxide and hydrogen in the presence of a catalyst such as cobalt octacarbonyl or a rhodium complex.[ 11]
H2 C=CH2 + CO + H2 → CH3 CH2 CH=O
CH3 CH2 CH=O + H2 → CH3 CH2 CH2 OH
Other proposed industrial processes include the two-step isomerization and hydrogenation of propylene oxide.[ 12]
A traditional laboratory preparation of 1-propanol involves treating n -propyl iodide with moist Ag2 O .
Safety
1-Propanol is thought to be similar to ethanol in its effects on the human body, but 2 to 4 times more potent according to a study conducted on rabbits. Many toxicology studies find oral acute LD50 ranging from 1.9 g/kg to 6.5 g/kg (compared to 7.06 g/kg for ethanol). It is metabolized into propionic acid . Effects include alcoholic intoxication and high anion gap metabolic acidosis. As of 2011, one case of lethal poisoning was reported following oral ingestion of 500mL of 1-propanol.[ 13]
1-Propanol as fuel
1-Propanol has a high octane number and is suitable for use as engine fuel. However, propanol is too expensive to use as a motor fuel. The research octane number (RON) of propanol is 118, and the anti-knock index (AKI) is 108.[ 14]
References
^ Favre HA, Powell WH (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 (Blue Book) . Cambridge: The Royal Society of Chemistry. p. 61. doi :10.1039/9781849733069 . ISBN 9780854041824 . ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0533" . National Institute for Occupational Safety and Health (NIOSH). ^ Pal A, Gaba R (2008). "Volumetric, acoustic, and viscometric studies of molecular interactions in binary mixtures of dipropylene glycol dimethyl ether with 1-alkanols at 298.15 K". J. Chem. Thermodyn. 40 (5): 818– 828. Bibcode:2008JChTh..40..818P . doi :10.1016/j.jct.2008.01.008 . ^ "n-Propyl alcohol" . Immediately Dangerous to Life or Health Concentrations . National Institute for Occupational Safety and Health.^ Chisholm, Hugh, ed. (1911). "Propyl Alcohols" Encyclopædia Britannica ^ Wisniak, Jaime (2013). "Gustav Charles Bonaventure Chancel" . Educación Química . 24 (1): 23– 30. doi :10.1016/S0187-893X(13)73191-4 ISSN 0187-893X . ^ s:fr:Page:Comptes rendus hebdomadaires des séances de l’Académie des sciences, tome 037, 1853.djvu/414 ^ Linnemann, Eduard (1868). "Ueber die Darstellung der Fettalkohole aus ihren Anfangsgliedern. Reduction des Essigsäure-Anhydrids zu Aethylalkohol" . Justus Liebigs Annalen der Chemie (in German). 148 (2): 249– 251. doi :10.1002/jlac.18681480216 . ISSN 1099-0690 . ^ Zeitschrift für Chemie ^ Schorlemmer, C. (1869). "On the Derivatives of Propane (Hydride of Propyl)" . Proceedings of the Royal Society of London . 17 : 372– 376. doi :10.1098/rspl.1868.0072 . ISSN 0370-1662 . ^
Papa AJ (2011). "Propanols". Ullmann's Encyclopedia of Industrial Chemistry . Weinheim: Wiley-VCH. doi :10.1002/14356007.a22_173.pub2 . ISBN 9783527303854 .
^ Papa, Anthony J. (2000). Ullmann's Encyclopedia of Industrial Chemistry 243– 254. doi :10.1002/14356007 . ISBN 978-3-527-30385-4 . ^ Unmack JL (2011). "N-PROPANOL Health-Base Assessment and Recommendation for HEAC" (PDF) . ^ "Bioalcohols" . Biofuel.org.uk . 2010. Retrieved 16 Apr 2014 .
Further reading
Furniss, B. S.; Hannaford, A. J.; Smith, P. W. G.; Tatchell, A. R. (1989), Vogel's Textbook of Practical Organic Chemistry (5th ed.), Harlow: Longman, ISBN 0-582-46236-3 Lide DR, ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). TF-CRC. ISBN 0849304873 . O'Neil MJ, ed. (2006). The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (14th ed.). Merck. ISBN 091191000X . Perkin WH, Kipping FS (1922). Organic Chemistry . London: W. & R. Chambers. ISBN 0080223540 .
External links
Alcohols
By consumption
Alcohols found in
1-Propanol 2-Methyl-1-butanol
Ethanol Isoamyl alcohol
Isobutanol
Phenethyl alcohol
tert -Amyl alcoholtert -Butyl alcoholTryptophol Medical alcohol
Ethchlorvynol
Methylpentynol
Methanol poisoning
Toxic alcohols
Primary
Methanol
4-Methylcyclohexanemethanol
Aminomethanol
Cyclohexylmethanol
Methoxymethanol
Methylazoxymethanol
Trifluoromethanol Ethanol
1-Aminoethanol
2,2,2-Trichloroethanol
2,2,2-Trifluoroethanol
2-(2-Ethoxyethoxy)ethanol
2-(2-Methoxyethoxy)ethanol
2-(2-Methoxyethoxy)ethanol
2-Butoxyethanol
2-Chloroethanol
2-Ethoxyethanol
2-Fluoroethanol
2-Mercaptoethanol
2-Methoxyethanol
Aminoethylethanolamine
Diethylethanolamine
Dimethylethanolamine
Ethanol Ethanolamine
N ,N -DiisopropylaminoethanolN -MethylethanolaminePhenoxyethanol
Tribromoethanol Butanol Straight-chain1 — C9 Straight-chain10 — C19
1-Decanol (capric)
1-Undecanol (hendecyl)
1-Dodecanol (lauryl)
1-Tridecanol
1-Tetradecanol (myristyl)
1-Pentadecanol
1-Hexadecanol (cetyl / palmityl)
1-Heptadecanol
1-Octadecanol (stearyl)
1-Nonadecanol Straight-chain20 — C29
1-Icosanol (arachidyl)
1-Heneicosanol
1-Docosanol (behenyl)
1-Tricosanol
1-Tetracosanol (lignoceryl)
1-Pentacosanol
1-Hexacosanol (ceryl)
1-Heptacosanol
1-Octacosanol (cluytyl / montanyl)
1-Nonacosanol Straight-chain30 — C39
1-Triacontanol (melissyl / myricyl)
1-Hentriacontanol
1-Dotriacontanol (lacceryl)
1-Tritriacontanol
1-Tetratriacontanol (geddyl)
1-Pentatriacontanol
1-Hexatriacontanol
1-Heptatriacontanol
1-Octatriacontanol
1-Nonatriacontanol Straight-chain40 — C49
1-Tetracontanol
1-Hentetracontanol
1-Dotetracontanol
1-Tritetracontanol
1-Tetratetracontanol
1-Pentatetracontanol
1-Hexatetracontanol
1-Heptatetracontanol
1-Octatetracontanol
1-Nonatetracontanol
2-Ethylhexanol
Allyl alcohol
Anisyl alcohol
Benzyl alcohol
Cinnamyl alcohol
Crotyl alcohol
Furfuryl alcohol
Isoamyl alcohol
Neopentyl alcohol
Nicotinyl alcohol
Perillyl alcohol
Phenethyl alcohol
Prenol
Propargyl alcohol
Salicyl alcohol
Tryptophol
Vanillyl alcohol
Veratrole alcohol Secondary
1-Phenylethanol
2-Butanol
2-Deoxyerythritol
2-Heptanol
3-Heptanol
2-Hexanol
3-Hexanol
3-Methyl-2-butanol
2-Nonanol
2-Octanol
2-Pentanol
3-Pentanol
Cyclohexanol
Cyclopentanol
Cyclopropanol
Diphenylmethanol
Isopropanol Pinacolyl alcohol
Pirkle's alcohol
Propylene glycol methyl ether
Tertiary
2-Methyl-2-pentanol
2-Methylheptan-2-ol
2-Methylhexan-2-ol
3-Methyl-3-pentanol
3-Methyloctan-3-ol
Diacetone alcohol
Ethchlorvynol
Methylpentynol
Nonafluoro-tert -butyl alcohol
tert -Amyl alcoholtert -Butyl alcoholTriphenylethanol
Triphenylmethanol Hydric alcohols
Monohydric alcohols Dihydric alcohols Trihydric alcohols Polyhydric alcohols (sugar alcohols)
Amyl alcohols
2,2-Dimethylpropan-1-ol
2-Methylbutan-1-ol
2-Methylbutan-2-ol
3-Methylbutan-1-ol
3-Methylbutan-2-ol
Pentan-1-ol
Pentan-2-ol
Pentan-3-ol Aromatic alcohols
Benzyl alcohol
2,4-Dichlorobenzyl alcohol
3-Nitrobenzyl alcohol Saturated
Cetostearyl alcohol
Decanol
Lauryl alcohol
Myristyl alcohol
Nonanol
Octanol
Tridecanol
Undecanol Branched and
3-Methyl-3-pentanol
Erucyl alcohol
Linolenyl alcohol
Linoleyl alcohol
Oleyl alcohol
Palmitoleyl alcohol
tert-Amyl alcohol
tert-Butyl alcohol Sugar alcohols
C1 — C7
Methylene glycol (C1 )
Ethylene glycol (C2 )Glycerol (C3 )Erythritol (C4 )
Threitol (C4 )
Arabitol (C5 )
Ribitol (C5 )
Xylitol (C5 )
Mannitol (C6 )
Sorbitol (C6 )Galactitol (C6 )
Iditol (C6 )
Volemitol (C7 ) Deoxy sugar Cyclic sugar Glycylglycitols
Maltitol
Lactitol
Isomalt
Maltotriitol
Maltotetraitol
Polyglycitol
Terpene alcohols
Monoterpene
Borneol
Citronellol
Geraniol
Linalool
Menthol Nerol
Rhodinol
Terpineol Sesquiterpene
Bisabolol
Farnesol
Nerolidol
Patchoulol Diterpene
Dialcohols Trialcohols Sterols
Cholesterol Ergosterol
Lanosterol
β-Sitosterol
Stigmasterol Fluoroalcohols
1,3-Difluoro-2-propanol
2,2,2-Trifluoroethanol
2-Fluoroethanol
Nonafluoro-tert -butyl alcohol
Trifluoromethanol Preparations Reactions
Acridine derivatives
Ethacridine lactate
9-Aminoacridine
Euflavine Biguanides and amidines
Dibrompropamidine
Chlorhexidine#
Propamidine
Hexamidine
Polihexanide Phenol and derivatives
Hexachlorophene
Policresulen
Phenol Triclosan
Triclocarban
Chloroxylenol#
Biphenylol
Fenticlor Nitrofuran derivatives Iodine products
Iodine/octylphenoxypolyglycolether
Povidone-iodine#
Diiodohydroxypropane Quinoline derivatives
Dequalinium
Chlorquinaldol
Oxyquinoline
Clioquinol Quaternary ammonium compounds
Benzalkonium
Benzethonium chloride
Cetrimonium (bromide/chloride)
Cetylpyridinium
Cetrimide
Benzoxonium chloride
Didecyldimethylammonium chloride Mercurial products
Mercuric amidochloride
Phenylmercuric borate
Mercuric chloride
Merbromin
Nitromersol
Thiomersal
Mercuric iodide Silver compoundsAlcohols Other
# WHO-EM‡ Withdrawn from marketClinical trials :
† Phase III§ Never to phase III
GABAA
GABAB
1,4-Butanediol 4-Fluorophenibut
Aceburic acid
Baclofen
GABOB
GHB (sodium oxybate)GBL
GVL
Phenibut
Tolibut
Valiloxybate (XW-10172) H1
α2 -Adrenergic
Clonidine Detomidine
Dexmedetomidine
Lofexidine
Medetomidine
Romifidine
Tasipimidine
Tizanidine
Xylazine 5-HT2A
Melatonin
Agomelatine
Melatonin
Ramelteon
Tasimelteon Orexin
Daridorexant
Lemborexant
Suvorexant α2 δ VDCC
Atagabalin
Gabapentin Gabapentin enacarbil
Mirogabalin
Phenibut
Pregabalin Others
Cannabinoids (e.g., cannabis , tetrahydrocannabinol /dronabinol, nabilone, cannabidiol, Zenivol)Chlorophenylalkyldiols
Fenpentadiol
Metaglycodol
Phenaglycodol
Diethylpropanediol
Evoxine
Fenadiazole
GABA reuptake inhibitors (e.g., tiagabine)
Guaifenesin-related muscle relaxants
Chlorphenesin
Mephenesin
Mephenoxalone
Metaxalone
Methocarbamol
Midaflur
Opioids (e.g., morphine )Passion flower Phemerazole
Scopolamine
Serotonin precursors (e.g., tryptophan , 5-hydroxytryptophan (5-HTP; oxitriptan))
Trazodone UMB68
Valnoctamide
GABA A receptor positive modulators
Alcohols
Brometone
Butanol Chloralodol
Chlorobutanol (cloretone)
Ethanol (alcohol) (alcoholic drink )
Ethchlorvynol
Isobutanol
Isopropanol Menthol Methanol Methylpentynol
Pentanol
Petrichloral
Propanol tert -Butanol (2M2P)tert -Pentanol (2M2B)Tribromoethanol
Trichloroethanol
Triclofos
Trifluoroethanol Barbiturates
(-)-DMBB
Allobarbital
Alphenal
Amobarbital
Aprobarbital
Barbexaclone
Barbital
Benzobarbital
Benzylbutylbarbiturate
Brallobarbital
Brophebarbital
Butabarbital/Secbutabarbital
Butalbital
Buthalital
Butobarbital
Butallylonal
Carbubarb
Crotylbarbital
Cyclobarbital
Cyclopentobarbital
Difebarbamate
Enallylpropymal
Ethallobarbital
Eterobarb
Febarbamate
Heptabarb
Heptobarbital
Hexethal
Hexobarbital
Metharbital
Methitural
Methohexital
Methylphenobarbital
Narcobarbital
Nealbarbital
Pentobarbital
Phenallymal
Phenobarbital Phetharbital
Primidone
Probarbital
Propallylonal
Propylbarbital
Proxibarbital
Reposal
Secobarbital
Sigmodal
Spirobarbital
Talbutal
Tetrabamate
Tetrabarbital
Thialbarbital
Thiamylal
Thiobarbital
Thiobutabarbital
Thiopental Thiotetrabarbital
Valofane
Vinbarbital
Vinylbital Benzodiazepines Carbamates
Carisbamate
Carisoprodol
Clocental
Cyclarbamate
Difebarbamate
Emylcamate
Ethinamate
Febarbamate
Felbamate
Hexapropymate
Hydroxyphenamate
Lorbamate
Mebutamate
Meprobamate
Nisobamate
Pentabamate
Phenprobamate
Procymate
Styramate
Tetrabamate
Tybamate Flavonoids
6-Methylapigenin
Ampelopsin (dihydromyricetin)
Apigenin
Baicalein
Baicalin
Catechin
EGC
EGCG
Hispidulin
Linarin
Luteolin
Rc-OMe
Skullcap constituents (e.g., baicalin)
Wogonin Imidazoles
Etomidate
Metomidate
Methoxyetomidate
Propoxate
Isopropoxate
Butomidate
Iso-butomidate
Sec-butomidate
CF2-Etomidate
CF3-Etomidate
CF3-Propoxate
Flutomidate
2,6-Dichloro-3-fluoroetomidate Kava constituents
10-Methoxyyangonin
11-Methoxyyangonin
11-Hydroxyyangonin
Desmethoxyyangonin
11-Methoxy-12-hydroxydehydrokavain
7,8-Dihydroyangonin
Kavain
5-Hydroxykavain
5,6-Dihydroyangonin
7,8-Dihydrokavain
5,6,7,8-Tetrahydroyangonin
5,6-Dehydromethysticin
Methysticin
7,8-Dihydromethysticin
Yangonin Monoureides
Acecarbromal
Apronal (apronalide)
Bromisoval
Carbromal
Capuride
Ectylurea Neuroactive steroids
Acebrochol
Allopregnanolone (brexanolone)
Alfadolone
Alfaxalone
3α-Androstanediol
Androstenol
Androsterone
Certain anabolic-androgenic steroids
Cholesterol DHDOC
3α-DHP
5α-DHP
5β-DHP
DHT
Etiocholanolone
Ganaxolone
Hydroxydione
Minaxolone
ORG-20599
ORG-21465
P1-185
Posovolone
Pregnanolone (eltanolone)
Progesterone Renanolone
SAGE-105
SAGE-324
SAGE-516
SAGE-689
SAGE-872
Testosterone THDOC
Zuranolone Nonbenzodiazepines
Cyclopyrrolones : EszopiclonePagoclone
Pazinaclone
Suproclone
Suriclone
Zopiclone Imidazopyridines : AlpidemDS-1
Necopidem
Saripidem
Zolpidem Pyrazolopyrimidines : DivaplonFasiplon
Indiplon
Lorediplon
Ocinaplon
Panadiplon
Taniplon
Zaleplon Others : AdipiplonCGS-8216
CGS-9896
CGS-13767
CGS-20625
CL-218,872
CP-615,003
CTP-354
ELB-139
GBLD-345
Imepitoin
JM-1232
L-838,417
Lirequinil (Ro41-3696)
Miltirone (rosmariquinone)
NS-2664
NS-2710
NS-11394
Pipequaline
ROD-188
RWJ-51204
SB-205,384
SX-3228
TGSC01AA
TP-003
TPA-023
TP-13
U-89843A
U-90042
Viqualine
Y-23684 Phenols
Cipepofol
Fospropofol
Propofol Propofol hemisuccinate
Thymol Piperidinediones
Glutethimide
Methyprylon
Piperidione
Pyrithyldione Pyrazolopyridines
Cartazolate
Etazolate
ICI-190,622
Tracazolate Quinazolinones
Afloqualone
Cloroqualone
Diproqualone
Etaqualone
Mebroqualone
Mecloqualone
Methaqualone Methylmethaqualone
Nitromethaqualone
SL-164 Volatiles/gases Others/unsorted
3-Hydroxybutanal
α-EMTBL
AA-29504
Alogabat
Avermectins (e.g., ivermectin)
Bromide compounds (e.g., lithium bromide, potassium bromide, sodium bromide)
Carbamazepine
Chloralose
Chlormezanone
Clomethiazole
Darigabat
DEABL
Deuterated etifoxine
Dihydroergolines (e.g., dihydroergocryptine, dihydroergosine, dihydroergotamine, ergoloid (dihydroergotoxine))
DS2
Efavirenz
Etazepine
Etifoxine
Fenamates (e.g., flufenamic acid, mefenamic acid, niflumic acid, tolfenamic acid)
Fluoxetine Flupirtine
Hopantenic acid
KRM-II-81
Lanthanum Lavender oil
Lignans (e.g., 4-O-methylhonokiol, honokiol, magnolol, obovatol)
Loreclezole
Menthyl isovalerate (validolum)
Monastrol
Nicotinic acid Nicotinamide
Org 25,435
Phenytoin
Propanidid
Retigabine (ezogabine)
Safranal
Seproxetine
Stiripentol
Sulfonylalkanes (e.g., sulfonmethane (sulfonal), tetronal, trional)
Terpenoids (e.g., borneol)
Topiramate
Valerian constituents (e.g., isovaleric acid, isovaleramide, valerenic acid, valerenol)Unsorted benzodiazepine site positive modulators: α-PineneMRK-409 (MK-0343)
TCS-1105
TCS-1205 See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators