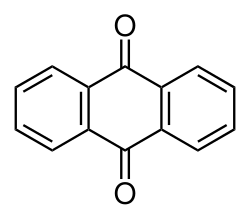
9,10-Anthraquinone[ 1]
Names
Preferred IUPAC name
Other names
Anthraquinone 9,10-Anthracenedione Anthradione 9,10-Anthrachinon Anthracene-9,10-quinone 9,10-Dihydro-9,10-dioxoanthracene Hoelite Morkit Corbit
Identifiers
3D model (JSmol)
Beilstein Reference
390030
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard
100.001.408
EC Number
Gmelin Reference
102870
KEGG
RTECS number
UNII
UN number
3143
InChI=1S/C14H8O2/c15-13-9-5-1-2-6-10(9)14(16)12-8-4-3-7-11(12)13/h1-8H
Key: RZVHIXYEVGDQDX-UHFFFAOYSA-N
O=C1c2ccccc2C(=O)c3ccccc13
Properties
C 14 H 8 O 2
Molar mass
−1
Appearance
Yellow solid
Density
1.438 g/cm3 [ 1]
Melting point
284.8 °C (544.6 °F; 558.0 K)[ 1]
Boiling point
377 °C (711 °F; 650 K)[ 1]
Solubility in water
Insoluble
Hazards
Occupational safety and health
Main hazards
possible carcinogen
GHS labelling:
Pictograms
Signal word
Danger
Hazard statements
H350
Precautionary statements
P201 , P202 , P281 , P308+P313 , P405 , P501
Flash point
185 °C (365 °F; 458 K)
Related compounds
Related compounds
quinone,anthracene
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Anthraquinone , also called anthracenedione or dioxoanthracene , is an aromatic organic compound with formula C14 8 2 . Several isomers exist but these terms usually refer to 9,10-anthraquinone (IUPAC : 9,10-dioxoanthracene) wherein the keto groups are located on the central ring. It is used as a digester additive to wood pulp for papermaking. Many anthraquinone derivatives are generated by organisms or synthesised industrially for use as dyes, pharmaceuticals, and catalysts . Anthraquinone is a yellow, highly crystalline solid, poorly soluble in water but soluble in hot organic solvents. It is almost completely insoluble in ethanol near room temperature but 2.25 g will dissolve in 100 g of boiling ethanol. It is found in nature as the rare mineral hoelite.
Synthesis
There are several current industrial methods to produce 9,10-anthraquinone:
The oxidation of anthracene . Chromium(VI) is the typical oxidant.
The Friedel–Crafts reaction of benzene and phthalic anhydride in presence of AlCl3 . o-Benzoylbenzoic acid is an intermediate. This reaction is useful for producing substituted anthraquinones.
The Diels-Alder reaction of naphthoquinone and butadiene followed by oxidative dehydrogenation.
The acid-catalyzed dimerization of styrene to give a 1,3-diphenylbutene, which then can be transformed to the anthraquinone.[ 3] BASF . It also arises via the Rickert–Alder reaction, a retro-Diels–Alder reaction.
Reactions
Hydrogenation gives dihydroanthraquinone (anthrahydroquinone). Reduction with copper gives anthrone.[ 4] [ 5] [ 6]
Applications
Digester additive in papermaking
9,10-Anthraquinone is used as a digester additive in production of paper pulp by alkaline processes, like the kraft, the alkaline sulfite or the Soda-AQ processes. The anthraquinone is a redox catalyst . The reaction mechanism may involve single electron transfer (SET).[ 7] cellulose and hemicellulose, and thereby protecting it from alkaline degradation (peeling). The anthraquinone is reduced to 9,10-dihydroxyanthracene which then can react with lignin . The lignin is degraded and becomes more watersoluble and thereby more easy to wash away from the pulp, while the anthraquinone is regenerated. This process gives an increase in yield of pulp, typically 1–3% and a reduction in kappa number.[ 8]
Hydrogen peroxide production
2-Alkyl-9,10-Anthroquinones are used as a catalyst in the anthraquinone process for the production of hydrogen peroxide. This process is the dominant industrial method of hydrogen peroxide production.[ 9]
Niche uses
9,10-anthraquinone is used as a bird repellant on seeds, and as a gas generator in satellite balloons.[ 10] kea attacks in New Zealand.[ 11]
Other isomers
Several other isomers of anthraquinone exist, including the 1,2-, 1,4-, and 2,6-anthraquinones. They are of minor importance compared to 9,10-anthraquinone.
Safety
Anthraquinone has no recorded LD50 , probably because it is so insoluble in water.
In terms of metabolism of substituted anthraquinones, the enzyme encoded by the gene UGT1A8 has glucuronidase activity with many substrates including anthraquinones.[ 12]
See also
Benzoquinone
Naphthoquinone
Parietin
2-Ethylanthraquinone
References
^ a b c d Haynes, William M., ed. (2016). CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. p. 3.28. ISBN 9781498754293 . ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 . The Royal Society of Chemistry. p. 724. doi :10.1039/9781849733069 . ISBN 978-0-85404-182-4 .^ Vogel, A. "Anthraquinone". Ullmann's Encyclopedia of Industrial Chemistry . Weinheim: Wiley-VCH. doi :10.1002/14356007.a02_347 . ISBN 978-3-527-30673-2 . ^ Macleod, L. C.; Allen, C. F. H. (1934). "Benzanthrone". Organic Syntheses . 14 : 4. doi :10.15227/orgsyn.014.0004 . ^ Scott, W. J.; Allen, C. F. H. (1938). "Potassium Anthraquinone-α-Sulfonate". Organic Syntheses . 18 : 72. doi :10.15227/orgsyn.018.0072 . ^ Scott, W. J.; Allen, C. F. H. (1938). "α-Chloroanthraquinone". Organic Syntheses . 18 : 15. doi :10.15227/orgsyn.018.0015 . ^ Samp, J. C. (2008). A comprehensive mechanism for anthraquinone mass transfer in alkaline pulping (Thesis). Georgia Institute of Technology. p. 30. hdl:1853/24767 . ^ Sturgeoff, L. G.; Pitl, Y. (1997) [1993]. "Low Kappa Pulping without Capital Investment". In Goyal, G. C. (ed.). Anthraquinone Pulping . TAPPI Press. pp. 3– 9. ISBN 0-89852-340-0 . ^ Campos-Martin, Jose M.; Blanco-Brieva, Gema; Fierro, Jose L. G. (2006). "Hydrogen Peroxide Synthesis: An Outlook beyond the Anthraquinone Process" Angewandte Chemie International Edition . 45 (42): 6962– 6984. doi :10.1002/anie.200503779 . ISSN 1521-3773 . ^ "www.americanheritage.com" . Archived from the original on 2009-06-09. Retrieved 2009-09-22 .^ Dudding, Adam (29 July 2012). "How to solve a problem like a kea" . Sunday Star Times . New Zealand. Retrieved 11 November 2014 . ^ Ritter, J. K.; Chen, F.; Sheen, Y. Y.; Tran, H. M.; Kimura, S.; Yeatman, M. T.; Owens, I. S. (1992). "A Novel Complex Locus UGT1 Encodes Human Bilirubin, Phenol, and other UDP-Glucuronosyltransferase Isozymes with Identical Carboxyl Termini" (PDF) . Journal of Biological Chemistry . 267 (5): 3257– 3261. doi :10.1016/S0021-9258(19)50724-4 PMID 1339448 .
External links
Authority control databases
International Other
Topics in organic reactions
A value
Alpha effect
Annulene
Anomeric effect
Antiaromaticity
Aromatic ring current
Aromaticity Baird's rule
Baker–Nathan effect
Baldwin's rules
Bema Hapothle
Beta-silicon effect
Bicycloaromaticity
Bredt's rule
Bürgi–Dunitz angle
Catalytic resonance theory
Charge remote fragmentation
Charge-transfer complex
Clar's rule
Conformational isomerism
Conjugated system
Conrotatory and disrotatory
Curtin–Hammett principle
Dynamic binding (chemistry)
Edwards equation
Effective molarity
Electromeric effect
Electron-rich
Electron-withdrawing group
Electronic effect
Electrophile
Evelyn effect
Flippin–Lodge angle
Free-energy relationship
Grunwald–Winstein equation
Hammett acidity function
Hammett equation
George S. Hammond
Hammond's postulate
Homoaromaticity
Hückel's rule
Hyperconjugation
Inductive effect
Kinetic isotope effect
LFER solvent coefficients (data page)
Marcus theory
Markovnikov's rule
Möbius aromaticity
Möbius–Hückel concept
More O'Ferrall–Jencks plot
Negative hyperconjugation
Neighbouring group participation
2-Norbornyl cation
Nucleophile
Kennedy J. P. Orton
Passive binding
Phosphaethynolate
Polar effect
Polyfluorene
Ring strain
Σ-aromaticity Spherical aromaticity
Spiroaromaticity
Steric effects
Superaromaticity
Swain–Lupton equation
Taft equation
Thorpe–Ingold effect
Vinylogy
Walsh diagram
Woodward–Hoffmann rules
Woodward's rules
Y-aromaticity Yukawa–Tsuno equation
Zaitsev's rule
Σ-bishomoaromaticity List of organic reactions
Carbon-carbon Homologation reactions
Arndt–Eistert reaction
Hooker reaction
Kiliani–Fischer synthesis
Kowalski ester homologation
Methoxymethylenetriphenylphosphorane
Seyferth–Gilbert homologation
Wittig reaction Olefination reactions
Bamford–Stevens reaction
Barton–Kellogg reaction
Boord olefin synthesis
Chugaev elimination
Cope reaction
Corey–Winter olefin synthesis
Dehydrohalogenation
Elimination reaction Grieco elimination
Hofmann elimination
Horner–Wadsworth–Emmons reaction
Hydrazone iodination
Julia olefination
Julia–Kocienski olefination
Kauffmann olefination
McMurry reaction
Peterson olefination
Ramberg–Bäcklund reaction
Shapiro reaction
Takai olefination
Wittig reaction
Carbon-heteroatom
Azo coupling
Bartoli indole synthesis
Boudouard reaction
Cadogan–Sundberg indole synthesis
Diazonium compound
Esterification Grignard reagent
Haloform reaction
Hegedus indole synthesis
Hurd–Mori 1,2,3-thiadiazole synthesis
Kharasch–Sosnovsky reaction
Knorr pyrrole synthesis
Leimgruber–Batcho indole synthesis
Mukaiyama hydration
Nenitzescu indole synthesis
Oxymercuration reaction
Reed reaction
Schotten–Baumann reaction
Ullmann condensation
Williamson ether synthesis
Yamaguchi esterification Degradation
Barbier–Wieland degradation
Bergmann degradation
Edman degradation
Emde degradation
Gallagher–Hollander degradation
Hofmann rearrangement
Hooker reaction
Isosaccharinic acid
Marker degradation
Ruff degradation
Strecker degradation
Von Braun amide degradation
Weerman degradation
Wohl degradation Organic redox
Acyloin condensation
Adkins–Peterson reaction
Akabori amino-acid reaction
Alcohol oxidation
Algar–Flynn–Oyamada reaction
Amide reduction
Andrussow process
Angeli–Rimini reaction
Aromatization
Autoxidation
Baeyer–Villiger oxidation
Barton–McCombie deoxygenation
Bechamp reduction
Benkeser reaction
Bergmann degradation
Birch reduction
Bohn–Schmidt reaction
Bosch reaction
Bouveault–Blanc reduction
Boyland–Sims oxidation
Cannizzaro reaction
Carbonyl reduction
Clemmensen reduction
Collins oxidation
Corey–Itsuno reduction
Corey–Kim oxidation
Corey–Winter olefin synthesis
Criegee oxidation
Dakin oxidation
Davis oxidation
Deoxygenation
Dess–Martin oxidation
DNA oxidation
Elbs persulfate oxidation
Emde degradation
Eschweiler–Clarke reaction
Étard reaction
Fischer–Tropsch process Fleming–Tamao oxidation
Fukuyama reduction
Ganem oxidation
Glycol cleavage
Griesbaum coozonolysis
Grundmann aldehyde synthesis
Haloform reaction
Hydrogenation Hydrogenolysis
Hydroxylation
Jones oxidation
Kiliani–Fischer synthesis
Kolbe electrolysis
Kornblum oxidation
Kornblum–DeLaMare rearrangement
Leuckart reaction
Ley oxidation
Lindgren oxidation
Lipid peroxidation
Lombardo methylenation
Luche reduction
Markó–Lam deoxygenation
McFadyen–Stevens reaction
Meerwein–Ponndorf–Verley reduction
Methionine sulfoxide
Miyaura borylation
Mozingo reduction
Noyori asymmetric hydrogenation
Omega oxidation
Oppenauer oxidation
Oxygen rebound mechanism
Ozonolysis
Parikh–Doering oxidation
Pinnick oxidation
Prévost reaction
Reduction of nitro compounds
Reductive amination
Riley oxidation
Rosenmund reduction
Rubottom oxidation
Sabatier reaction
Sarett oxidation
Selenoxide elimination
Shapiro reaction
Sharpless asymmetric dihydroxylation
Epoxidation of allylic alcohols
Sharpless epoxidation
Sharpless oxyamination
Stahl oxidation
Staudinger reaction
Stephen aldehyde synthesis
Swern oxidation
Transfer hydrogenation
Wacker process
Wharton reaction
Whiting reaction
Wohl–Aue reaction
Wolff–Kishner reduction
Wolffenstein–Böters reaction
Zinin reaction Rearrangement
1,2-rearrangement
1,2-Wittig rearrangement
2,3-sigmatropic rearrangement
2,3-Wittig rearrangement
Achmatowicz reaction
Alkyne zipper reaction
Allen–Millar–Trippett rearrangement
Allylic rearrangement
Alpha-ketol rearrangement
Amadori rearrangement
Arndt–Eistert reaction
Aza-Cope rearrangement
Baker–Venkataraman rearrangement
Bamberger rearrangement
Banert cascade
Beckmann rearrangement
Benzilic acid rearrangement
Bergman cyclization
Bergmann degradation
Boekelheide reaction
Brook rearrangement
Buchner ring expansion
Carroll rearrangement
Chan rearrangement
Claisen rearrangement
Cope rearrangement
Corey–Fuchs reaction
Cornforth rearrangement
Criegee rearrangement
Curtius rearrangement
Demjanov rearrangement
Di-π-methane rearrangement
Dimroth rearrangement
Divinylcyclopropane-cycloheptadiene rearrangement
Dowd–Beckwith ring-expansion reaction
Electrocyclic reaction
Ene reaction
Enyne metathesis
Favorskii reaction
Favorskii rearrangement
Ferrier carbocyclization
Ferrier rearrangement
Fischer–Hepp rearrangement
Fries rearrangement
Fritsch–Buttenberg–Wiechell rearrangement
Gabriel–Colman rearrangement
Group transfer reaction
Halogen dance rearrangement
Hayashi rearrangement
Hofmann rearrangement
Hofmann–Martius rearrangement
Ireland–Claisen rearrangement
Jacobsen rearrangement
Kornblum–DeLaMare rearrangement
Kowalski ester homologation
Lobry de Bruyn–Van Ekenstein transformation
Lossen rearrangement
McFadyen–Stevens reaction
McLafferty rearrangement
Meyer–Schuster rearrangement
Mislow–Evans rearrangement
Mumm rearrangement
Myers allene synthesis
Nazarov cyclization reaction
Neber rearrangement
Newman–Kwart rearrangement
Overman rearrangement
Oxy-Cope rearrangement
Pericyclic reaction
Piancatelli rearrangement
Pinacol rearrangement
Pummerer rearrangement
Ramberg–Bäcklund reaction
Ring expansion and contraction
Ring-closing metathesis
Rupe reaction
Schmidt reaction
Semipinacol rearrangement
Seyferth–Gilbert homologation
Sigmatropic reaction
Skattebøl rearrangement
Smiles rearrangement
Sommelet–Hauser rearrangement
Stevens rearrangement
Stieglitz rearrangement
Thermal rearrangement of aromatic hydrocarbons
Tiffeneau–Demjanov rearrangement
Vinylcyclopropane rearrangement
Wagner–Meerwein rearrangement
Wallach rearrangement
Weerman degradation
Westphalen–Lettré rearrangement
Willgerodt rearrangement
Wolff rearrangement Ring forming
1,3-Dipolar cycloaddition
Annulation
Azide-alkyne Huisgen cycloaddition
Baeyer–Emmerling indole synthesis
Bartoli indole synthesis
Bergman cyclization
Biginelli reaction
Bischler–Möhlau indole synthesis
Bischler–Napieralski reaction
Blum–Ittah aziridine synthesis
Bobbitt reaction
Bohlmann–Rahtz pyridine synthesis
Borsche–Drechsel cyclization
Bucherer carbazole synthesis
Bucherer–Bergs reaction
Cadogan–Sundberg indole synthesis
Camps quinoline synthesis
Chichibabin pyridine synthesis
Cook–Heilbron thiazole synthesis
Cycloaddition
Darzens reaction
Davis–Beirut reaction
De Kimpe aziridine synthesis
Debus–Radziszewski imidazole synthesis
Dieckmann condensation
Diels–Alder reaction Feist–Benary synthesis
Ferrario–Ackermann reaction
Fiesselmann thiophene synthesis
Fischer indole synthesis
Fischer oxazole synthesis
Friedländer synthesis
Gewald reaction
Graham reaction
Hantzsch pyridine synthesis
Hegedus indole synthesis
Hemetsberger indole synthesis
Hofmann–Löffler reaction
Hurd–Mori 1,2,3-thiadiazole synthesis
Iodolactonization
Isay reaction
Jacobsen epoxidation
Johnson–Corey–Chaykovsky reaction
Knorr pyrrole synthesis
Knorr quinoline synthesis
Kröhnke pyridine synthesis
Kulinkovich reaction
Larock indole synthesis
Madelung synthesis
Nazarov cyclization reaction
Nenitzescu indole synthesis
Niementowski quinazoline synthesis
Niementowski quinoline synthesis
Paal–Knorr synthesis
Paternò–Büchi reaction
Pechmann condensation
Petrenko-Kritschenko piperidone synthesis
Pictet–Spengler reaction
Pomeranz–Fritsch reaction
Prilezhaev reaction
Pschorr cyclization
Reissert indole synthesis
Ring-closing metathesis
Robinson annulation
Sharpless epoxidation
Simmons–Smith reaction
Skraup reaction
Urech hydantoin synthesis
Van Leusen reaction
Wenker synthesis Cycloaddition
1,3-Dipolar cycloaddition
4+4 Photocycloaddition
(4+3) cycloaddition
6+4 Cycloaddition
Alkyne trimerisation
Aza-Diels–Alder reaction
Azide-alkyne Huisgen cycloaddition
Bradsher cycloaddition
Cheletropic reaction
Conia-ene reaction
Cyclopropanation
Diazoalkane 1,3-dipolar cycloaddition
Diels–Alder reaction Enone–alkene cycloadditions
Hexadehydro Diels–Alder reaction
Intramolecular Diels–Alder cycloaddition
Inverse electron-demand Diels–Alder reaction
Ketene cycloaddition
McCormack reaction
Metal-centered cycloaddition reactions
Nitrone-olefin (3+2) cycloaddition
Oxo-Diels–Alder reaction
Ozonolysis
Pauson–Khand reaction
Povarov reaction
Prato reaction
Retro-Diels–Alder reaction
Staudinger synthesis
Trimethylenemethane cycloaddition
Vinylcyclopropane (5+2) cycloaddition
Wagner-Jauregg reaction Heterocycle forming reactions
Algar–Flynn–Oyamada reaction
Allan–Robinson reaction
Auwers synthesis
Bamberger triazine synthesis
Banert cascade
Barton–Zard reaction
Bernthsen acridine synthesis
Bischler–Napieralski reaction
Bobbitt reaction
Boger pyridine synthesis
Borsche–Drechsel cyclization
Bucherer carbazole synthesis
Bucherer–Bergs reaction
Chichibabin pyridine synthesis
Cook–Heilbron thiazole synthesis
Diazoalkane 1,3-dipolar cycloaddition
Einhorn–Brunner reaction
Erlenmeyer–Plöchl azlactone and amino-acid synthesis
Feist–Benary synthesis
Fischer oxazole synthesis
Gabriel–Colman rearrangement
Gewald reaction
Hantzsch ester
Hantzsch pyridine synthesis
Herz reaction
Knorr pyrrole synthesis
Kröhnke pyridine synthesis
Lectka enantioselective beta-lactam synthesis
Lehmstedt–Tanasescu reaction
Niementowski quinazoline synthesis
Nitrone-olefin (3+2) cycloaddition
Paal–Knorr synthesis
Pellizzari reaction
Pictet–Spengler reaction
Pomeranz–Fritsch reaction
Prilezhaev reaction
Robinson–Gabriel synthesis
Stollé synthesis
Urech hydantoin synthesis
Wenker synthesis
Wohl–Aue reaction